Visitors
Start Date Visitors: 24/04/2020
AMJAM is an open-access journal publishes Original article, Review article, Brief Research, Case report, Special article (Invited by Editor), Editorial, and Letter to the Editor in all health sciences, medical specialties and alternative medicine. AMJAM publishes 4 times a year in April (Issue 1), August (Issue 2), December (Issue 3), and 1 Supplementary issue. All submitted articles will be evaluated using double-blinded review process by 2-4 reviewers.
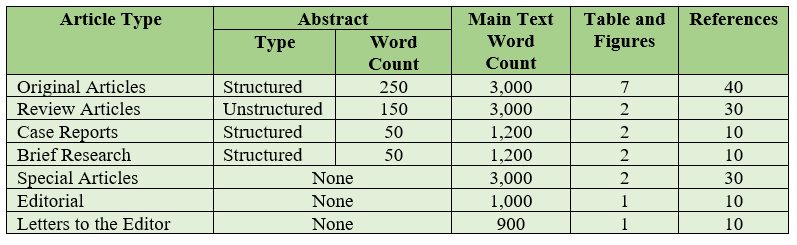
Original Article should include a title page, a structured abstract of no more than 250 words, a text of no more than 3,000 words, no more than 7 tables and figures, and no more than 40 references. Original Article Template
Review Article should include a title page, a narrative abstract of no more than 150 words, a text of no more than 3,000 words, no more than 2 tables or figures, and no more than 30 references. Review Article Template
Case Report should include a title page, a narrative abstract of no more than 50 words, a text of no more than 1,200 words, no more than 2 tables or figures, and no more than 10 references. Case Reports Template
Brief Research should include a title page, a narrative abstract of no more than 50 words, a text of no more than 1,200 words, no more than 2 tables or figures, and no more than 10 references. Brief Research Template
Special Article (Invited by Editor) should include a title page, a text of no more than 3,000 words, no more than 2 tables or figures, and no more than 30 references. Abstract nor keywords are not required. Special Article Template
Editorial should include a title page, a text of no more than 1,000 words, no more than 1 tables or figures, and no more than 10 references. Abstract nor keywords are not required. Editorial Template
Letter to the Editor should not exceed 900 words, no more than 1 table or figure, and no more than 10 references. Abstract nor keywords are not required. Letters to the Editor Template
The summarizes the requirements for different article types:
Commentaries are by invitation only. Please contact the journal office if you are interested in writing a Commentary.
The submission requires
1. Title Page and Manuscript
2. Copy of Ethic Approval
3. Graphical Abstract
MANUSCRIPT PREPARATION
Authors who are not fluent in English should have their manuscript checked by a native speaker of English and/or an editing service that provides such assistance. Please concept editorial office if author would like to receive suggestion on editing service. Manuscripts that do not follow the required format or are poorly prepared may be asked to revised for resubmission. In the submitted cover letter author should state that "All authors have significantly contributed to the research."
All submitted manuscript must include institutional ethical approval certificate. Single space the entire manuscript, including title page, abstract, body, references, tables, and figure legends. Use left justification only, so that the right margin is ragged. Number pages consecutively, beginning with the title page. Use a standard font (such as Times New Roman or Helvetica) and set the font size to 12 points (for tables as well as text). All numbers published in AMJAM will be in Arabic numbers. Each component of the article should begin on a separate page, as follows: title page, abstract, body text, acknowledgments, references, appendices, tables and figure legends. All these components must be in a single file.
Title Page
The title page should include the following information: (1) the title of the manuscript; (2) the names of the author(s), including each author’s highest academic degree or professional certification; (3) the departmental and institutional affiliation of each author, including city, state, and country; (4) the name, address, telephone number, fax number, and e-mail address of the author responsible for correspondence, and (if different) the name and address to be used for reprint requests; (5) if relevant, a statement about any previous presentation of the data or findings in a preliminary report or abstract; (6) an abbreviated title of not more than 45 characters (including spaces), to be used as a running head in print and for searching results online; and (7) a word count for the body of the text (ie, excluding the abstract and the references). Acknowledgment of financial support and potential conflicts of interest must be included and should be placed in the Acknowledgments section (see below).
Abstract
Original Article should include a structured abstract of no more than 250 words. The following headings are suggested: Introduction/Objective, Design, Methods (or Interventions), Results, Conclusions, and Key words (3-5 words or phrases). If this list of headings is inappropriate, variations are permitted: for example, a study that involved no intervention would use the heading “Methods” rather than “Intervention”; or an analysis of an existing data set might use the heading “Methods” in place of both “Intervention”. For brevity, parts of the abstract can be written in phrases rather than complete sentences (eg, “Design: Retrospective cohort study” or “Design: Before-after trial”).
The contents of each section should conform to the guidelines below.
Body Text
The main sections and subdivisions of the body text should be indicated by side heads flushed with the left margin and two lines above the text. Keep Methods, Results, and Discussion distinct and separate. The Methods section should provide detail sufficient to allow others to reproduce your experiment. Methods may not be described or restated in figure legends or table notes, but must be all together in the Methods section. The Results section contains the previously unpublished data derived by this application of your methods, without commentary (beyond the minimum that might be necessary to ensure intelligibility to the reader). The Discussion section contains your interpretation of the reported data and comments on its meaning. There should be no separate section labeled “Conclusion.” Avoid duplicating in the text data that have been provided in tables or figures (minimal duplication, for emphasis or clarity, is acceptable). Also avoid duplication within the text; for example, the Discussion section should not restate all the findings that have been presented in Results and/or in tables and figures.
Acknowledgments
Financial support. Information on financial support should be provided by authors. The Acknowledgments section should list all sources of financial support for the work, including any financial arrangement with a company whose product is related to the study. If there was no financial support, that too should be stated. Acknowledgments, including grant support, should be placed after the text.
Example:
Financial support. The XXX Project is supported by the Thai Ministry of Health. Additional support for this study was provided by Becton-Dickinson.
Financial support. None reported.
Conflict of interest. The Acknowledgments section must contain a statement of potential conflicts of interest. If the manuscript is accepted for publication, the disclosures will be published. The Acknowledgments section of the manuscript must list the name of each contributing author and any potential conflicts of interest for each author for the previous three years; if no potential conflict exists, that too should be stated.
Example:
Potential conflicts of interest. K.L.H. reports having consulted for and having received grant support from Astellas and reports having received an honorarium from Cubist before starting employment with the New York Department of Public Health in 2009.
Potential conflicts of interest. All authors report no conflicts of interest relevant to this article.
References
Use the Style Guide of the American Medical Association (AMA) as a reference. References should be cited consecutively in the text, with superscript numbers placed outside periods and commas and inside colons and semicolons. References cited only in tables or figure legends should be numbered as though all were cited at the point at which the table or figure was first mentioned.
A paper that is “in press” may be included in the reference list if it has been accepted for publication. Citations such as “in preparation,” “submitted for publication,” “unpublished data,” and “personal communication” should be given in parentheses in the text only, including the names of all individuals to whom the information should be attributed, as well as each person’s highest academic degree and the month and year of the information’s origin. For personal communications, specify whether the communication was written or oral.
At the end of each manuscript, list the references in numerical order, single spaced, according to the order they are cited in the text. If there are 7 or more authors, list the first 3 authors’ names, followed by “et al”; otherwise, list all authors. Abbreviations of journal names should conform to Index Medicus or MEDLINE. Unlisted journals should not be abbreviated. Authors are responsible for bibliographic accuracy. Journal titles should be cited as they existed at the time of publication. Format references according to the style given in the AMA Manual of Style, 10th Edition.
AMA CITATION STYLE GUIDE
Journal article (examples)
Pittet D, Simon A, Hugonnet S, Pessoa-Silva CL, Sauvan V, Perneger TV. Hand hygiene among physicians: performance, beliefs, and perceptions. Ann Intern Med. 2004;141:
1-8.
Camins BC, Richmond AM, Dyer KL, et al. A crossover intervention trial evaluating the efficacy of a chlorhexidine-impregnated sponge in reducing catheter-related bloodstream infections among patients undergoing hemodialysis. Infect Control Hosp Epidemiol. 2010;31:1118-1123.
Journal article in press (example)
Figueroa P, Johanssen KL, Price FG, et al. Outbreak of Acinetobacter infection in a neonatal intensive care unit. Pediatr Infect Dis J (in press).
Paper presented at a professional meeting (example)
Chen LF, Freeman JT, Sexton DJ, Choi YI, Anderson DJ. NHSN definition of laboratory-detected BSI is overly sensitive for Enterococcus. In: Program and abstracts of the 19th Annual Scientific Meeting of the Society for Healthcare Epidemiology of America (SHEA); March 18–22, 2009; San Diego, CA. Abstract 359.
Book (example)
Heoprich PD. Infectious Diseases. 2nd ed. New York, NY: Harper & Row; 1977.
Chapter in a book (example)
Schaffner W. Psittacosis: ornithosis, parrot fever. In: Beeson PB, McDermott W, Wyngaarden JB, eds. Cecil Textbook of Medicine. 15th ed. Philadelphia, PA: W. B. Saunders; 1979:336-338.
Web page (example)
7. Clinical laboratory fee schedule. Centers for Medicare and Medicaid Services
website. https://www.cms.gov/ClinicalLabFeeSched/02_clinlab.asp#TopOfPage.
Published 2010. Accessed April 2, 2010.
Tables & Figure
Tables and figures in articles are clear and well configured within article content (are not just copied-pasted from Excel)
Tables
Prepare tables with the MS Word table editor; text formatted to look like a table by use of tabs and hard returns is not acceptable and will be rejected. Include tables in the same file of the manuscript, not in separate files. Tables should be single spaced. Number tables in the order in which they are cited in the text, and provide a descriptive title for each table.
Every column in a table requires a head that describes the contents of the cells below. The units of measure for all data must be clearly stated in the heads, in the stub (leftmost) column, or in data cells, as appropriate. Do not use vertical lines, and do not use ditto marks for repeated information.
List and define any abbreviations in a note below the table, above the table footnotes (no footnote designator is required for this line), even if the abbreviations have been defined in the text. Use superscript letters for footnote designators.
The tables that are too large to be reproduced in print, if accepted for publication, will appear only in the online version of the article, and information about the online-only table (including a full or partial title) will be included in the print version of the article.
Examples of Table:
Figure
- Size: Image with a minimum of 35 x 14 cm. (w x h) using a minimum resolution of 300 dpi. Larger image, please use the same ratio.
- File types: PDF or MS Office files
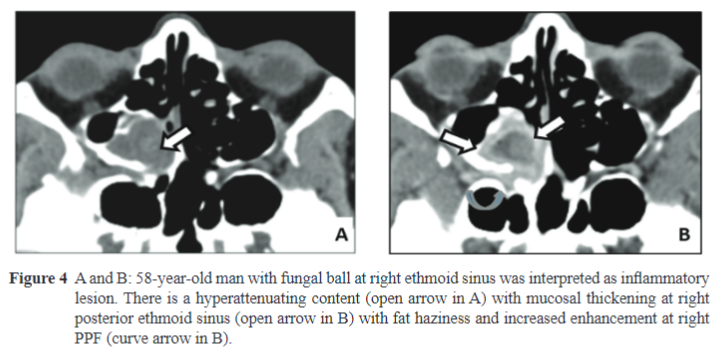
Examples of Figure:
GRAPHICAL ABSTRACT
Authors can consider provide an original image that clearly represents the work described in the manuscript. Graphical abstracts should be submitted as a separate file in the submission system.
Size: Image with a minimum of 35 x 14 cm. (w x h) using a minimum resolution of 300 dpi. Larger image, please use the same ratio.
Font: Times New Roman
File types: PDF or MS Office files
WITHDRAWING A MANUSCRIPT
To withdraw your manuscript after reviewing by the reviewer (but not published), the authors must write a clear and concise letter with explanation as to why the manuscript needs to be withdrawn. The letter must be signed by the author.
